COVID-19 vaccine developed in Thailand can be stored in refrigerator for three months

[ad_1]
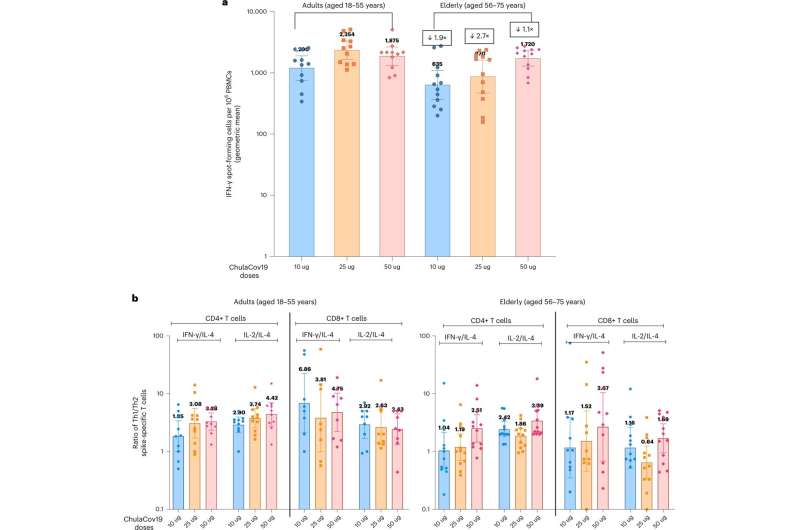
A crew of researchers affiliated with a number of entities in Thailand, working with two colleagues from the U.S. and two from Canada, has developed a mRNA COVID-19 vaccine that may be safely refrigerated for as much as three months earlier than use. The crew has named it ChulaCov19.
Of their paper revealed within the journal Nature Microbiology, the group describes the variations between their vaccine and different mRNA COVID-19 vaccines.
Amid the worldwide pandemic, teams world wide have developed vaccines to guard or reduce signs of individuals from/with COVID-19. And of the 172 vaccines developed to this point, 40 are RNA-based. Essentially the most well-known vaccines have been developed by Pfizer and Moderna, and each have been proven to be efficient in stopping critical signs.
Each vaccines will also be saved in a fridge, although their most storage time is vastly completely different. The Pfizer vaccine will be saved for 10 weeks and the Moderna vaccine for simply 30 days. For longer intervals, each should be frozen at very chilly temperatures, Pfizer at -60°C and -15°C for Moderna.
These necessities symbolize important hurdles to be used in third-world nations. On this new effort, the researchers have prolonged refrigeration occasions to 3 months with their vaccine.
The brand new vaccine was made in the identical primary method because the Moderna and Pfizer vaccines: mRNA is used to induce the physique into producing COVID-19 spike proteins. It additionally makes use of a lipid to cowl the mRNA, however makes use of a unique one than both Moderna or Pfizer—it doesn’t break down as rapidly in a fridge.
Additionally, the brand new vaccine will not be prefusion stabilized, a course of that helps the spike proteins take form contained in the physique. Testing of the brand new vaccine confirmed that it was simply as efficient as the opposite two vaccines with out the stabilization.
Testing discovered it to be efficient in opposition to the delta, gamma, beta and alpha variants, although it was discovered to be much less efficient in opposition to the omicron variant. Additionally, unwanted side effects from the vaccine have been just like these discovered with the opposite two vaccines. The brand new vaccine is at present in a Part II scientific trial.
Sivaporn Gatechompol et al, Security and immunogenicity of a prefusion non-stabilized spike protein mRNA COVID-19 vaccine: a part I trial, Nature Microbiology (2022). DOI: 10.1038/s41564-022-01271-0
© 2022 Science X Community
Quotation:
COVID-19 vaccine developed in Thailand will be saved in fridge for 3 months (2022, November 19)
retrieved 20 November 2022
from https://medicalxpress.com/information/2022-11-covid-vaccine-thailand-refrigerator-months.html
This doc is topic to copyright. Aside from any truthful dealing for the aim of personal examine or analysis, no
half could also be reproduced with out the written permission. The content material is supplied for info functions solely.
[ad_2]




